Section 2: Application of Personalized Medicine - Pharmacogenomics
The Current Medication Dilemma
When a person has an illness, disease, or any medical problem that can be treated with medication, doctors often prescribed the medication that is known to be most beneficial and have the most efficacy for patients. In order to determine and confirm the effectiveness of these popular and commonly-prescribed drugs, the drugs, which are created by pharmaceutical companies, must go through extensive clinical trials and studies for approval. Then, doctors can prescribe them to patients, and patients can take the drugs for treatment. However, even though a drug may be considered popular and effective for many people who have used it to treat Disease X, this does not necessarily mean that the medication is perfect for every individual who is sick with this disease. Often, many individuals experience severe side-effects of popular medications, and these are the types of dramatic side-effects we hear about in commercial advertisements for different medications, where the narrator mentions something along the lines of this: "Drug X has been known to cause various mild-to-severe side effects, including..." There are hundreds of thousands of medications that have been created over time, but the true problem lies in the fact that sometimes, individuals are prescribed medications that can cause discomfort, harm, or overall lack of improvement when all the patient wants is relief. Is there a way to make sure patients are prescribed medications that will be less harmful, cause less (or less severe) side effects, and ultimately be more effective in treating various illnesses that a person may have? This is where pharmacogenomics, or pharmaceutical genomics, comes into play.
When a person has an illness, disease, or any medical problem that can be treated with medication, doctors often prescribed the medication that is known to be most beneficial and have the most efficacy for patients. In order to determine and confirm the effectiveness of these popular and commonly-prescribed drugs, the drugs, which are created by pharmaceutical companies, must go through extensive clinical trials and studies for approval. Then, doctors can prescribe them to patients, and patients can take the drugs for treatment. However, even though a drug may be considered popular and effective for many people who have used it to treat Disease X, this does not necessarily mean that the medication is perfect for every individual who is sick with this disease. Often, many individuals experience severe side-effects of popular medications, and these are the types of dramatic side-effects we hear about in commercial advertisements for different medications, where the narrator mentions something along the lines of this: "Drug X has been known to cause various mild-to-severe side effects, including..." There are hundreds of thousands of medications that have been created over time, but the true problem lies in the fact that sometimes, individuals are prescribed medications that can cause discomfort, harm, or overall lack of improvement when all the patient wants is relief. Is there a way to make sure patients are prescribed medications that will be less harmful, cause less (or less severe) side effects, and ultimately be more effective in treating various illnesses that a person may have? This is where pharmacogenomics, or pharmaceutical genomics, comes into play.
Pharmacogenomics: Making Medicine Just for You
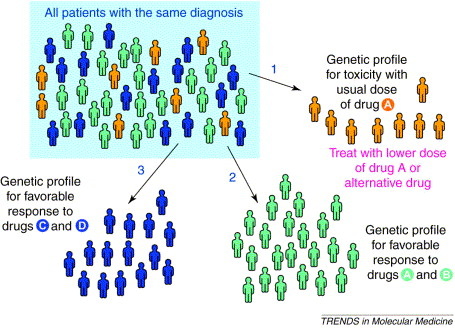
In order to approach the problem with medications that either do not work for a patient or cause side-effects that are too severe for continued use, scientists and doctors have turned to genetics. Although many factors, like age, race, gender, and drug combinations, can contribute to variability in drug success among patients with different diseases, studies have determined that there is also a connection between genetic make-up and variability in patient responses to prescription drugs (1). This establishes the goal of pharmacogenomics. The ultimate goal is to improve the outcome of drug therapies for all patients (1) by gathering genetic information from individuals that could provide insight on how patients would respond to or metabolize various drugs. Ultimately, pharmacogenomics aims to make the best patient-drug treatment match possible.
Scientists first focus on gathering information about the various genes, including drug-metabolizing genes, that interact with medications that a patient takes. By collecting information about the variation in these sequences among different individuals, scientists can determine the relationship between specific sequences for these genes and the predicted way a patient would react to taking a specific medication for a certain disease. The variation in these critical sequences are what contribute to variable drug response among individuals of a population (1), helping to explain why your parent might respond well to using one medication and see improvement in their condition, while your parent's friend might not see the same improvement. Most of the analytical work in this area has been conducted on single genes, but as you may or may not recall from your professor's lecture on genetics and gene expression, the physical attributes of a person, or their phenotype, when it comes to side-effects and drug response variability, are likely caused by multiple genes interacting (1).
There are typically two drug treatment approaches: trial and error, and the per protocol approach (1). In the trial and error approach, doctors prescribe patients drug after drug until the "perfect" match between patient and medication is created. With the per protocol approach, patients with certain cancers, heart failures, and post-transplantation patients are treated almost exactly the same with predetermined treatment strategies commonly used to help with that condition (1). As you can guess, both of these methods result in wasted time of patient and practitioner, wasted money by patient and insurance companies, and incredible levels of frustration when treatment options do not prove to be effective. By utilizing pharmacogenomics, we can essentially eliminate these issues and hopefully increase the success rate of drug treatments for most individuals over time. With pharmacogenomics, we can predict how a patients gene sequence will affect his or her uptake of medication and how his or her body will react over time, attempting to narrow in on treatment options that will maximize health benefits.
In order to approach the problem with medications that either do not work for a patient or cause side-effects that are too severe for continued use, scientists and doctors have turned to genetics. Although many factors, like age, race, gender, and drug combinations, can contribute to variability in drug success among patients with different diseases, studies have determined that there is also a connection between genetic make-up and variability in patient responses to prescription drugs (1). This establishes the goal of pharmacogenomics. The ultimate goal is to improve the outcome of drug therapies for all patients (1) by gathering genetic information from individuals that could provide insight on how patients would respond to or metabolize various drugs. Ultimately, pharmacogenomics aims to make the best patient-drug treatment match possible.
Scientists first focus on gathering information about the various genes, including drug-metabolizing genes, that interact with medications that a patient takes. By collecting information about the variation in these sequences among different individuals, scientists can determine the relationship between specific sequences for these genes and the predicted way a patient would react to taking a specific medication for a certain disease. The variation in these critical sequences are what contribute to variable drug response among individuals of a population (1), helping to explain why your parent might respond well to using one medication and see improvement in their condition, while your parent's friend might not see the same improvement. Most of the analytical work in this area has been conducted on single genes, but as you may or may not recall from your professor's lecture on genetics and gene expression, the physical attributes of a person, or their phenotype, when it comes to side-effects and drug response variability, are likely caused by multiple genes interacting (1).
There are typically two drug treatment approaches: trial and error, and the per protocol approach (1). In the trial and error approach, doctors prescribe patients drug after drug until the "perfect" match between patient and medication is created. With the per protocol approach, patients with certain cancers, heart failures, and post-transplantation patients are treated almost exactly the same with predetermined treatment strategies commonly used to help with that condition (1). As you can guess, both of these methods result in wasted time of patient and practitioner, wasted money by patient and insurance companies, and incredible levels of frustration when treatment options do not prove to be effective. By utilizing pharmacogenomics, we can essentially eliminate these issues and hopefully increase the success rate of drug treatments for most individuals over time. With pharmacogenomics, we can predict how a patients gene sequence will affect his or her uptake of medication and how his or her body will react over time, attempting to narrow in on treatment options that will maximize health benefits.
The Economic Advantage of Personalized Medicine through Pharmacogenomics
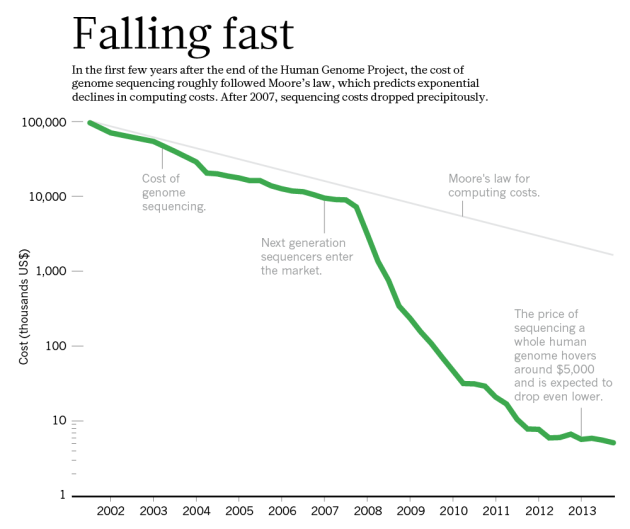
Pharmacogenomics can ultimately improve patient health and safety while improving health care costs and efficiency at the same time, which would both be incredibly beneficial in our society and others. The United States has an annual healthcare cost per individual per year (for 2010 to 2014) of approximately $8,895 (3), and compared to other countries, that's pretty high! The idea is that if we can make genome sequencing less expensive, this can encourage more people to have their whole genome sequenced, and with an analysis of a patient's sequence by genetic experts, doctors can utilize genetic information to make specialized treatment plans for patients. Maybe if we tackle medical problems from a genetic standpoint, we can decrease the overall healthcare cost per individual in the long run! There are companies with the knowledge and technological instruments capable of sequencing your Average Joe's entire genome, which is roughly 3 billion base-pairs long. However, the cost to complete this task is not small. In 2014, the estimated cost for genome sequencing hovered around $5,000 (4) but this cost is dramatically lower than the starting cost when the first human genome was sequenced, which cost around $100,000,000 at the turn of the 21st century (4). The cost is expected to continually decrease each year, potentially making whole genome sequencing financially possible for the average American within the next decade! While pharmacogenomic testing and research are a bit pricey right now, due to the cost of specific genetic tests, screenings, labor, equipment, and data analysis, the goal is for these costs associated with genome sequencing and genetic analyses to decrease over time so more individuals can utilize the power of pharmacogenomics in disease and illness treatment.
Pharmacogenomics can ultimately improve patient health and safety while improving health care costs and efficiency at the same time, which would both be incredibly beneficial in our society and others. The United States has an annual healthcare cost per individual per year (for 2010 to 2014) of approximately $8,895 (3), and compared to other countries, that's pretty high! The idea is that if we can make genome sequencing less expensive, this can encourage more people to have their whole genome sequenced, and with an analysis of a patient's sequence by genetic experts, doctors can utilize genetic information to make specialized treatment plans for patients. Maybe if we tackle medical problems from a genetic standpoint, we can decrease the overall healthcare cost per individual in the long run! There are companies with the knowledge and technological instruments capable of sequencing your Average Joe's entire genome, which is roughly 3 billion base-pairs long. However, the cost to complete this task is not small. In 2014, the estimated cost for genome sequencing hovered around $5,000 (4) but this cost is dramatically lower than the starting cost when the first human genome was sequenced, which cost around $100,000,000 at the turn of the 21st century (4). The cost is expected to continually decrease each year, potentially making whole genome sequencing financially possible for the average American within the next decade! While pharmacogenomic testing and research are a bit pricey right now, due to the cost of specific genetic tests, screenings, labor, equipment, and data analysis, the goal is for these costs associated with genome sequencing and genetic analyses to decrease over time so more individuals can utilize the power of pharmacogenomics in disease and illness treatment.
Is Pharmacogenomics Already Being Implemented in Healthcare?
Although pharmacogenomics is a relatively new field, there are already applications of this technology and information in the medical field today. For example, with individuals infected with Human Immunodeficiency Virus (HIV), the individual may be prescribed an antiviral drug abacavir (Ziagen), but through many clinical trials, it was discovered that the drug causes hypersensitivity characterized by fever, rash, nausea, and extreme fatigue (5). Because more African Americans experience the hypersensitivity than Caucasians, researchers suspected that there was a genetic component determining hypersensitivity towards the drug (6). Research lead to the discovery of a significant association between a specific allele of the human leukocyte antigen (HLA) B*5701 (yes, that's a complicated name, but do not worry about that!) and the appearance of the hypersensitivity symptoms! It has been found that individuals with a variation in this allele are likely to experience the hypersensitivity symptoms, and the package insert for Zaigen suggests screening for this allele before taking the medication (5). In addition to its application to antiretroviral medications and HIV treatment, pharmacogenomics can be useful in genetic screening for variants in genes that cause negative side effects from use of a specific breast cancer drug treatment called Herceptin and with an acute lymphoblastic leukemia chemotherapy drug called Purinethol (5). Some patients taking Purinethol have a genetic variant that negatively impacts their ability to synthesize and process the drug in their bodies, causing severe side effects (5). As you can see, pharmacogenomics is already proving to be useful in the medical field for selective drug treatment of patients with certain illnesses, and if genome sequencing becomes more common and available, the number of disease treatments that could benefit from pharmacogenomics will increase.
Although pharmacogenomics is a relatively new field, there are already applications of this technology and information in the medical field today. For example, with individuals infected with Human Immunodeficiency Virus (HIV), the individual may be prescribed an antiviral drug abacavir (Ziagen), but through many clinical trials, it was discovered that the drug causes hypersensitivity characterized by fever, rash, nausea, and extreme fatigue (5). Because more African Americans experience the hypersensitivity than Caucasians, researchers suspected that there was a genetic component determining hypersensitivity towards the drug (6). Research lead to the discovery of a significant association between a specific allele of the human leukocyte antigen (HLA) B*5701 (yes, that's a complicated name, but do not worry about that!) and the appearance of the hypersensitivity symptoms! It has been found that individuals with a variation in this allele are likely to experience the hypersensitivity symptoms, and the package insert for Zaigen suggests screening for this allele before taking the medication (5). In addition to its application to antiretroviral medications and HIV treatment, pharmacogenomics can be useful in genetic screening for variants in genes that cause negative side effects from use of a specific breast cancer drug treatment called Herceptin and with an acute lymphoblastic leukemia chemotherapy drug called Purinethol (5). Some patients taking Purinethol have a genetic variant that negatively impacts their ability to synthesize and process the drug in their bodies, causing severe side effects (5). As you can see, pharmacogenomics is already proving to be useful in the medical field for selective drug treatment of patients with certain illnesses, and if genome sequencing becomes more common and available, the number of disease treatments that could benefit from pharmacogenomics will increase.
Interactive Walkthrough of Pharmacogenomics for a Cancer Patient
As mentioned, pharamacogenomics is a relatively new and quickly-developing field! Genetics studies and how genes relate to the variability in drug response among individuals is currently being used in treatment for various types of cancers, including colorectal cancer and acute lyphoblastic leukemia. The following interactive walk-through will take you through the role of pharmacogenomics in the treatment of a young girl with leukemia so you may see the benefits of this field in a real-world example!
As mentioned, pharamacogenomics is a relatively new and quickly-developing field! Genetics studies and how genes relate to the variability in drug response among individuals is currently being used in treatment for various types of cancers, including colorectal cancer and acute lyphoblastic leukemia. The following interactive walk-through will take you through the role of pharmacogenomics in the treatment of a young girl with leukemia so you may see the benefits of this field in a real-world example!
A Huge Step in the Direction of Personalized Medicine (and Pharmacogenomics)
In the United States' 2015 State of the Union, President Barack Obama interesting took a minute of the address to propose personalized/precision medicine initiatives for the near future. He states that he wants our country to create "a new era of medicine - one that delivers the right treatment at the right time." He even made a huge funding request ($215 million) for tackling this project of making personalized medicine current, not something we image for the future. Listen to the snippet about precision medicine in the video below (29:06-29:55) and read about these initiatives on the Precision Medicine Initiative Factsheet.
In the United States' 2015 State of the Union, President Barack Obama interesting took a minute of the address to propose personalized/precision medicine initiatives for the near future. He states that he wants our country to create "a new era of medicine - one that delivers the right treatment at the right time." He even made a huge funding request ($215 million) for tackling this project of making personalized medicine current, not something we image for the future. Listen to the snippet about precision medicine in the video below (29:06-29:55) and read about these initiatives on the Precision Medicine Initiative Factsheet.
Discussion Questions
1) What do you think are some of the problems keeping individuals from wanting to participate in genome sequencing that is necessary for pharmacogenomics to be implemented on a wider scale? Explain.
2) How could we convince more people to participate in necessary genome sequencing to make personalized medicine and pharmacogenomics more widely utilized in the healthcare field?
3) Personal Opinion Question: Would you like to have your genome sequenced for medical purposes? Why or why not? Discuss with entire class.
1) What do you think are some of the problems keeping individuals from wanting to participate in genome sequencing that is necessary for pharmacogenomics to be implemented on a wider scale? Explain.
2) How could we convince more people to participate in necessary genome sequencing to make personalized medicine and pharmacogenomics more widely utilized in the healthcare field?
3) Personal Opinion Question: Would you like to have your genome sequenced for medical purposes? Why or why not? Discuss with entire class.
Sources
1) Johnson JA (November 2003). "Pharmacogenetics: potential for individualized drug therapy through genetics.". Trends Genet 19 (11): 660–6. DOI 10.1016/j.tig.2003.09.008
http://www.sciencedirect.com.prx.library.gatech.edu/science/article/pii/S0168952503002622#
2) http://www.cancer.net/navigating-cancer-care/how-cancer-treated/personalized-and-targeted-therapies/pharmacogenomics
3) http://data.worldbank.org/indicator/SH.XPD.PCAP
4) http://www.nature.com/news/technology-the-1-000-genome-1.14901
5) http://www.medscape.com/viewarticle/740288
6) Hetherington S, McGuirk S, Powell G, et al. Hypersensitivity reactions during therapy with the nucleoside reverse transcriptase inhibitor abacavir. Clin Ther. 2001;23:1603-1614.
1) Johnson JA (November 2003). "Pharmacogenetics: potential for individualized drug therapy through genetics.". Trends Genet 19 (11): 660–6. DOI 10.1016/j.tig.2003.09.008
http://www.sciencedirect.com.prx.library.gatech.edu/science/article/pii/S0168952503002622#
2) http://www.cancer.net/navigating-cancer-care/how-cancer-treated/personalized-and-targeted-therapies/pharmacogenomics
3) http://data.worldbank.org/indicator/SH.XPD.PCAP
4) http://www.nature.com/news/technology-the-1-000-genome-1.14901
5) http://www.medscape.com/viewarticle/740288
6) Hetherington S, McGuirk S, Powell G, et al. Hypersensitivity reactions during therapy with the nucleoside reverse transcriptase inhibitor abacavir. Clin Ther. 2001;23:1603-1614.